
Study of the impact and global role of RNA G-quadruplexes in transcriptome
| » Back to research |
|
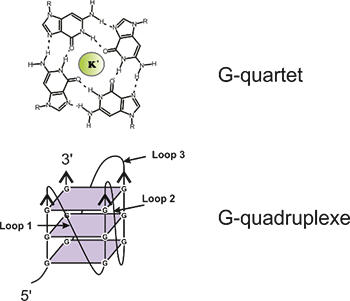
The life of an mRNA is dotted with several maturation and regulatory events. For a long time, it was generally accepted that the main way of regulating gene expression was at the transcriptional level. However, it has recently been reported that 90% of our genome is transcribed into RNA. Following this discovery, it is logical to think that posttranscriptional regulation could be a key piece of gene expression as well. The G-quadruplex structure is adopted by nucleic acid molecules rich in guanines. This structure is formed of a stack of "G-quartets" which are composed of 4 coplanar guanines and require the presence of monovalent metal cations, usually K +. The recognition of G-quadruplexes as regulatory elements found in the genome begins to be established little by little. However, the majority of research in this area focuses on G-quadruplex DNA and very little on those composed of RNA. |
|
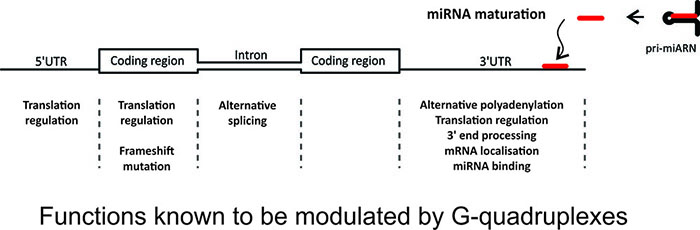
Depending on where they are in the pre-messenger RNA, G-quadruplexes can regulate translation, polyadenylation, alternative splicing and microRNA maturation. Some G-quadruplexes are also known to influence mRNA localization. |
|
Thanks to its 3 different phases (in silico, in vitro and in cellulo), our approach allowed us to evaluate the global role of G-quadruplexes of RNA in the cell, more precisely at the level of untranslated regions of mRNAs. Our research results allow the identification of new G-quadruplexes, notably through the use of artificial intelligence. By automated learning, we are now able to identify new G-quadruplexes in any genome. After identifying a G-quadruplex of interest, it is necessary to check whether the structure can be formed by in vitro assays on a synthetic RNA. We use a technique called "in line probing" which allows to highlight the formation of a G-quadruplex in different salt conditions. Biophysical methods such as circular dichroism and fluorescence methods using G-quadruplex specific fluorescent ligands are also used. Subsequently, we use luciferase assays in cells to demonstrate the effect of these G-quadruplexes on gene expression. The formation / deformation of G-quadruplexes thus makes it possible to control certain cellular mechanisms such as a switch. We have developed a technique that can promote or prevent the formation of a G-quadruplex in cell using specific oligonucleotides. More recently, we have begun studying the proteins that can bind G-quadruplexes and their effects on gene expression. As a result, RNA G-quadruplexes become potential new therapeutic targets. |
 |
|
